|
6/29/2023 0 Comments Workdone in thermodynamics nasa
Suddenly, without much of a transition, new pockets of industry arose, focusing towards large-scaled machines rather than small hand tools large industrial corporations often crushed small agriculturally centered commerce and in many areas, city life rendered country farm cottages obsolete. Prior to the mid-eighteenth century, the general European populace randomly dotted the land in small agricultural communities, industry was run out of country cottages, and scientific developments were nearly at a standstill. However, European society was about to experience unforeseeable rapid changes. Indeed, philosophy and science were inseparable in several emerging disciplines (this is always true of new fields where no firm basis of study has yet been conducted). Science was viewed as purely a philosophic endeavor, where little research was conducted beyond the most useful fields. Society prior to the eighteenth century favored developments in the life sciences (largely for medical research) and astronomy (for navigation and a record of the passage of time - also a source for early mythology and folklore). Our story begins back in the mid-seventeenth century. 
To give you a better understanding on how these laws came about and their modern scope of coverage, you have to understand when and why these laws were generated. However, since their conception, these laws have become some of the most important laws of all science - and are often associated with concepts far beyond what is directly stated in the wording. Basically, the First Law of Thermodynamics is a statement of the conservation of energy - the Second Law is a statement about the direction of that conservation - and the Third Law is a statement about reaching Absolute Zero (0' K). In simplest terms, the Laws of Thermodynamics dictate the specifics for the movement of heat and work. You cannot get out of the game (because absolute zero is unattainable).Īnswered by: Dan Summons, Physics Undergrad Student, UOS, Souhampton.You cannot break even (you cannot return to the same energy state, because there isĪlways an increase in disorder entropy always increases).You cannot win (that is, you cannot get something for nothing, because matter and energy.Snow had an excellent way of remembering the three Thermodynamics is the study of the inter-relation between heat, work and internal energy of We start by discussing spontaneous processes and explain why some processes require work to occur even if energy would have been conserved.What is a simple defintion of the laws of thermodynamics? Asked by: James Beal Snow stating that the first law means “you can’t win.” He paraphrased the second law as “you can’t break even, except on a very cold day.” Unless you are at zero kelvin, you cannot convert 100% of thermal energy into work. In the chapter covering the first law of thermodynamics, we started our discussion with a joke by C. We cannot use internal energy stored in the air to propel a car, or use the energy of the ocean to run a ship, without disturbing something around that object. We cannot unmix cream from coffee without a chemical process that changes the physical characteristics of the system or its environment. 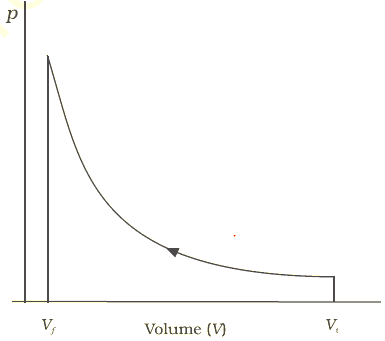
Energy cannot arbitrarily pass from one object to another, just as we cannot transfer heat from a cold object to a hot one without doing any work. One such principle is the second law of thermodynamics, which limits the use of energy within a source. 
So some other thermodynamic principles must be controlling the behavior of physical systems. For example, when two bodies are in thermal contact, heat never flows from the colder body to the warmer one, even though this is not forbidden by the first law. But this cannot be the only restriction imposed by nature, because many seemingly possible thermodynamic processes that would conserve energy do not occur. (credit: modification of work by NASA/JPL)Īccording to the first law of thermodynamics, the only processes that can occur are those that conserve energy. The ion propulsion engine is the first nonchemical propulsion to be used as the primary means of propelling a spacecraft. Introduction Figure 4.1 A xenon ion engine from the Jet Propulsion Laboratory shows the faint blue glow of charged atoms emitted from the engine.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
